 First, are there any structures possible where all the formal charges are zero? There are not, so we move on to rule #2. Given these calculated formal charges, let’s consult the guidelines discussed above. Next, the formal charge of the nitrogen in the middle: (5) – (½)(8) – (0) = +1.įinally, the formal charge of the oxygen: (6) – (½)(4) – (4) = 0.Īgain, first we calculate the formal charge of the nitrogen on the left: (5) – (½)(2) – (6) = -2.įinally, the formal charge of the oxygen: (6) – (½)(6) – (2) = +1. Similarly, we calculate the formal charge of the nitrogen on the left: (5) – (½)(4) – (4) = -1. Oxygen has 6 valence electrons, and this atom has 2 bonded electrons and 6 unbonded, thus the FC is (6) – (½)(2) – (6) = -1. This one has 8 bonded electrons and no unbonded, thus the FC is (5) – (½)(8) – (0) = +1.įinally, we calculate the formal charge of the oxygen. Next, we calculate it for the nitrogen in the middle. Nitrogen has 5 valence electrons, this atom has 6 bonded electrons (a triple bond), and 2 unbonded electrons, thus the formal charge is (5) – (½)(6) – (2) = 0. Let’s figure out which structure is correct.įirst, we calculate the formal charge of the nitrogen on the left.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


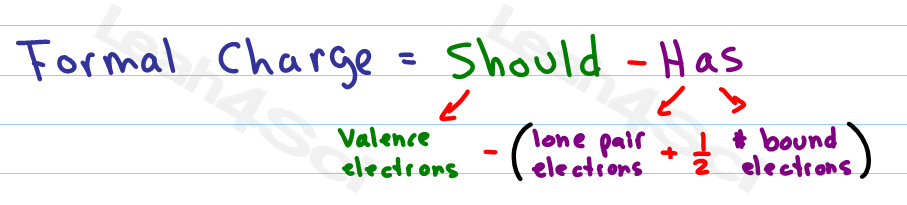

 RSS Feed
RSS Feed
